Are All Dark Glass Bottles used for Storing Extra Virgin Olive Oil the Same?
admin | April 25, 2010EVOO oxidises in two ways. One is called auto-oxidation and the other, and arguably the most destructive sort, is known as photo-oxidation. Photo-oxidation as its name suggests in initiated by light.
The good old fashioned oxygen that you find in air and that is dissolved in EVOO isn’t by itself very destructive. However, if it is hit by light which is packed with enough of an energetic punch, then it can push the generally harmless oxygen to a new and dangerous psychotic state. This energised oxygen, called triplet oxygen starts a destructive chain reaction, oxidising fat molecules left right and centre. The ultimate result of this reaction is a rancid oil that lacks fresh olive flavours, and worst still, fills your food with aweful flavours of old fish and chip shop or suburban corner grill.
Photo-oxidation can be simply avoided by ensuring that the oil is not exposed to light. To this end, good EVOO producers pack their oils in dark glass bottles or cans, and they implore their customers to store their oils in a dark place. While tin cans are completely impervious to light, dark glass is not. Different coloured glass of different levels of darkness are used to store EVOO. Look carefully: some are a classic green, others more of a “sea green” colour. Others are more olive in colour and yet others are amber.
Many producers think that the darker the glass the better, and that is all there is to it. Well they would be wrong.
Oxygen can only be energized if it is hit by light with a short enough wavelength – particularly light in the ultraviolet part of the spectrum. So, coloured glass that absorbs UV light will best protect the oil from oxidation. Those that let UV light through will not afford any protection to the oil, regardless of how dark or light the bottle appears to the eye.
So how do we measure the effectiveness of different glasses and material in blocking UV light? Scientists use a machine called a spectrophotometer or spectro for this gig. It sends a beam of continuously changing wavelength light through the glass and measures the amount of that light that doesn’t make it through (called the absorbance).
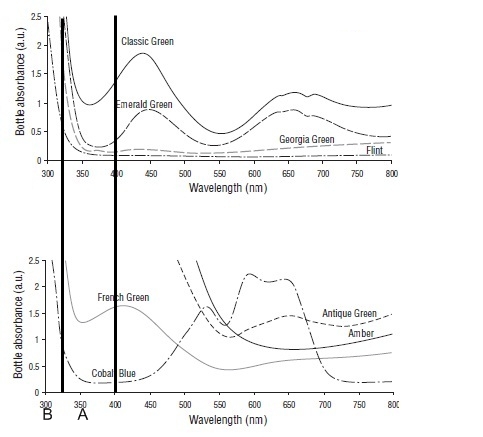
Each type of coloured bottle has a distinct ‘absorbance’ fingerprint. Some colours absorb light better at different wavelengths than others. Indeed, that is the exact reason why they are perceived by our eye as having different colours in the first place. The figure below shows the absorbance fingerprints of a variety of commonly used wine bottle colours (from Skouroumounis et al. 2006).
You’ll notice that there are a number of different types of green represented – classic green, antique green, emerald green and the like. They’re all green but they clearly absorb different wavelength light to different extents. The glass labeled ‘flint’ is better known to us as clear glass. It doesn’t absorb light at any wavelength. All the light just passes right through. Hence it looks clear and colourless.
But not all wavelengths are born equal when it comes to destroying EVOO. The ‘light’ with wavelength of 400 nanometers (nm) or less is known as ultraviolet radiation. Our eyes can’t see it but the spectro can. Below 315nm it is known as UV-B, and above 315 UV-A. (the dark lines on both figures represent these parts of the spectrum) .Both types have enough energy to excite oxygen and cause eventually cause rancidity. The absorbance at the other longer (visible) wavelengths gives the bottles their distinct colour.
So think about what the consequences would be of storing olive oil in cobalt blue bottles. They’ll look pretty funky (I guess), but cobalt blue glass doesn’t absorb very well in the UV part of the spectrum. So that means that any UV that hits the bottle will pass straight through the glass and attack the oil. And the glass will provide little protection regardless of how dark the blue. Darker blue glass will absorb more in the parts of the spectrum that don’t influence oxidation but will still let pretty well all the damaging UV light through.
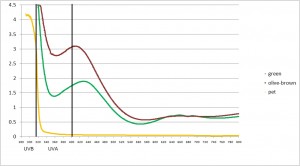
So what about olive oil bottles? This morning I intentionally smashed two common types of square bottles that are used to store EVOO. I bought the oils at a local specialty store. One bottle was common dark green and the other an equally common dark olive green/brown colour. I also cut a bit out of a clear PET bottle used to store olive oil that I purchased at a supermarket and measured the absorbance from the visible through to the UV part of the spectrum. Here’s what the glass looked like and below that are the results.
(Yellow=PET, Aqua=green glass, Brown=olive green/brown)
Clearly the olive/brown version is better than green when it comes to blocking UV. But neither is perfect. So store those oils in a dark place regardless of the fact that they are in dark glass. The PET was a bit of surprise. It absorbed very well in the UV-B, but was pretty well a clear window for UV-A. Some PET contains inbuilt UV blockers. This was clearly one of them. Not that you could tell by looking at it!








Hi Richard,
Great entry, as ever. I take it that you mean Skouroumounis et al, 2003? Do I correctly read the graph, and the abstract’s statement that “A420 of white wine in bottles of Antique Green or Amber glass could not be measured directly due to a strong absorbance of that wavelength by the glass” to indicate that these colors are nearly impervious to the critical range of UV? If so, shouldn’t these colors be the industry gold standard (lousy pun) — and how does a producer or a consumer know what “Antique Green” is?) Finally, what is the significance of the statement that “a strong correlation was established between measurement of A420 (cuvette) and A540 for white wine in Antique Green bottles and between A420 (cuvette) and either A540 or A600 for white wine in Amber bottles”?
Hm. Perhaps I should be asking the authors … Tell me to PO if appropriate 😉 . Thanks again!
Hi Michael
The paper I referred to was of course a wine paper, but it had some interesting information that was relevent to storing EVOO in bottles.
But as you asked… The purpose of the paper was to show that in some cases, the amount of browning of a white wine in a bottle of wine could be measured without opening it. Something pretty useful for QC. That abstract statement you referred to was stating that bottles of some colours like antique green absorb quite a bit at the wavelength (420nm) that are known to be good indicators of browning in white wines. Therefore for these coloured bottles it was not possible to reliably measure the amount of browning of the wine contained within them – well not using the traditional wavelength of 420nm.
Incidently 420nm is in the visible range of light so it isn’t that energetic as far as starting off rancidity reactions.
The last statement you refer to says… well if the glass interferes with our readings at 420nm we need to find another wavelength that could be an indicator of browning. What they found that the actual browning of the wine (as measured in a clear glass cuvette), correlated well with the 540 reading when in antique green. So by switching the measurement to 520 when measuring through antique green bottles did a good job of predicting white wine browning.
Lastly, you have to be in the trade to know exactly what the different colours are, but I’m sure any bottle rep would be happy to help if you were in the market for some – but just for now its that greeney turquoisey colour you often see used in expensive heavy wine bottles, and flint glass=clear glass.
RG
I would like to add a few comments regarding colored glasses for EVOO bottling.
Most glasses absorb in the UV range (have you tried getting a sun tan behind a glass window?). Only very expensive glass will transmit UV light, and that kind is used in the laboratory for absorbance measurements in that low range of the spectrum.
So what matters for the shelf life of oil is the absorbance on the higher side of the spectrum. Don’t forget that oil itself has color, and its pigments do absorb light in the visible side of the spectrum. Did you know that chlorophyll in plant cells also gets photo-oxidized. What about tocopherol?Both absorb light in different ranges of the visible spectrum.Thus colored glass is better than clear to filter light. And better yet, storage in the dark (no light, no photons) is always advised.
Photo oxydation of olive oil is thus easily avoidable It is chemical auto-oxidation the major culprit of oil rancidity. Hence, the best indicator of oil’s longevity and quality is very low free-acidity in the beginning, when milled and bottled.
Hi Liliana
Thanks for your considered comments. There’s no doubt that heat and oxygen are major enemies of olive oil. I’ve been considering writing about auto-oxidation for a whilel, but as you point out there are lots of facets to that story. I would point out that clear glass does transmit a good proportion of UV radiation. Various reports give its transmission at 72% (Tuchinda et al. 2006) and 74% (Duarte et al. 2009), and it’s this type of radiation that is energetic enough to excite chlorophyll resulting in its photo-oxidation.
RG
Hi Liliana,
You wrote:
Most glasses absorb in the UV range (have you tried getting a sun tan behind a glass window?).
No, but the level of UV energy required to acutely oxidize melanin leading to a compensatory increase in production is presumably much higher than the level required over a year of storage to gradually degrade olive oil, especially granted the ability of lipid oxidation products to propagate oxidative damage through chain reactions.
Don’t forget that oil itself has color, and its pigments do absorb light in the visible side of the spectrum. Did you know that chlorophyll in plant cells also gets photo-oxidized. What about tocopherol?Both absorb light in different ranges of the visible spectrum.
Yes, and those are bad things! Those pigments are things like carotenoids, which are antioxidants and valuable nutrients (as is tocopherol). And the breakdown (normally thermal) of chlorophyll into pyropheophytins is one of the indicators that the Aussies adopted as a surrogate marker for oxidized and/or adulterated oil.
Thus colored glass is better than clear to filter light. And better yet, storage in the dark (no light, no photons) is always advised.
Glad we agree …
Psomiadou and Tsimidou, 2002 find that, under the relatively severe condition of low indirect-sunlight-equivalent light storage, photooxidation led to “pheophytin a gradual degradation (>90%) accompanied by a considerable α-tocopherol loss (22−35%) due to the reaction of the latter with singlet oxygen. No changes were recorded for carotenoids, which acted as physical quenchers and light filters. Squalene loss was confined (4−12%). … Squalene was preferentially consumed to protect α-tocopherol.”