Thou shalt not store extra virgin olive oil in plastic bottles – Are you sure?
admin | August 6, 2012Extra Virgin Olive Oil has three basic commandments.
1. Thou shalt not store olive oil in the light
2. Thou shalt not store olive oil in a warm place
and
3. Thou shalt not store olive oil in plastic
Oxidation reactions in all fats including olive oil are catalysed by light. Once the reaction starts, it is self propagating and unstoppable. The oxidation reaction creates free radicals that oxidise fat molecules – a by product of which is more free radicals that go on to oxidise more fat molecules. The inevitable result is an avalanche of oxidation, and like nearly all chemical reactions in nature, the warmer the conditions the faster it goes. Thus explains the role of heat and light in a junior high chemistry sort of way.
But let’s move onto the purpose of this blog. To investigate the last commandment that ‘Though shalt not store olive oil in plastic’.
In short the story line goes like this: If you store olive oil in plastic containers then it will oxidise faster than if you store it in glass. I presume that the belief stems from the very reasonable tenet that glass is totally impermeable to oxygen but plastics are not. So of course it must be the case. But is it?
My interest in this matter was pricked, or more accurately rekindled by a powerpoint slide presented at a 2011 Australian EVOO conference by Claudia Guillaume from Modern Olives. Her work focussed on the changes during bottle storage of a couple of new chemical indices that had recently become part of the new Australian olive oil standard. But what raised my eyebrows was a rather innocuous figure on one of her slides that it appeared to show that an extra virgin olive oils stored in amber coloured PET plastic bottles outperformed glass with respect to some of the trusted measures of oxidation.
I had been tentatively looking at the scientific literature on the topic for a while but I always left feeling frustrated regarding the types of comparisons that were being made. There were many examples of ‘clear’ examples of one packaging (usually PET) being compared with ‘dark’ examples of another (always glass). Sometimes the oils were being stored seemingly endlessly in full light and under hot conditions presumably just to reinforce that heat and light are the enemies of extra virgin olive oil. Strange as no one was really questioning it.
In one notable instance, glass bottles were filled with extra virgin olive oil to a normal fill height, but the PET bottles were only filled to 1/3rd volume, the remaining 2/3rds of a bottle being full of apparently inert air. The PET containers were subjected to full light for two months, but those in glass were kept in the dark. The researchers concluded that “the quality loss of extra-virgin olive oil following storage in PET bottles for only 2 months was beyond the acceptable level from both the analytical and sensory points of view” (and incidentally they didn’t conduct any formal sensory trials). I wonder what they were expecting? Unfortunately, early papers in any field have a habit of cementing a collective persistent mind-set, particularly if the conclusions are in line with peoples’ expectations. In another instance (a very recent one I might add) the scientists put one extra virgin olive oil in glass and a different EV in PET. They didn’t mention that in their methods (you would think it an important detail wouldn’t you), but it was clear to see when you inspected the basic analysis of the oils at t=0 that they were different. The oils in PET had higher initial oxidation indices, so the avalanche was half completed in the PET bottles before the experiment had even begun.
The second reason for looking at the question more carefully is that advances in packaging technology have resulted in the wide commercial availability of many new variants of PET. Today, there are dark e.g. amber coloured, and totally opaque versions. They can be moulded with thicker walls and therefore lower oxygen permeability. Other technologies have succeeded in bringing down the oxygen permeability of PET v1.0 by between 2 and 7 fold (Gambacorta et al. 2004). The first called passive barrier technology involves placing additional thin barrier film(s) over the PET in order to make it harder for the oxygen molecules to pass through. The other, called active technology, involves either impregnating UV light blocking or oxygen scavenging substances into the molecular polymer matrix of the plastic. In the latter case, as oxygen permeates the PET, some of it gets chewed up by the impregnated antioxidants before it can damage the oil contained inside. Active and passive technologies have also been combined into a single polymer – sort of a oxygen condom and oxygenicide all in one package if you like.
But before we start. A little about the indices that people have used to measure oxidation and general oil quality in these studies.
Free Fatty Acidity FFA – measures the amount of oil degradation. Most degradation occurs before or at harvest due to enzymatic activity which can only occur in the olive and during processing. After an oil has been bottled enzymatic activity is pretty well zero so increases in FFA are usually small after bottling.
Peroxide Value (PV): is an index of the number of free radicals that are in the oil. As such it is a good measure of an oils potential to oxidise. It is a tricky one to interpret as it is very dynamic. Peroxides are formed by oxidation, but are in turn used up as the oil ages. So as an oil ages they can go up or down depending on what’s happening in the complex world of oxidation and reduction in the oil. So while a high PV is a very bad sign in a young oil, it could be argued that a ‘potential to oxidise’ measure applied to a 12 month old bottled oil is not of great practical importance.
K232: When an oil oxidises it forms compounds called dienes that absorb ultraviolet light strongly at the particular wavelength of 232 nanometres (nm). As it is a measure of how much an oil has oxidised (rather than its potential to do so), it is an important measure in the context of longer term packaging studies such as these.
K270: When an oil oxidises even further it forms compounds called trienes. These absorb strongly at the wavelength of 270 nm. So as per the explanation above, high K270 values mean that an oil is in an advanced stage of oxidation. This is another important measure of the amount of oxidation experienced by an oil at the end of its commercial shelf life.
Polyphenols are naturally occurring antioxidants that sacrifice themselves in a valiant attempts to fight oxidation, and longer and more intense the fight, the greater the reduction in polyphenols over the life of the oil.
In light of some of the less than satisfactory comparisons performed in the past, I scoured the literature to find published papers whereby they demonstrated some semblance of a fair comparison.The studies discussed met the following criteria:
1) the major experimental variables were properly controlled and replicated
and
2) logical and fair comparisons were made i.e.
– The same coloured containers (clear/clear or dark/dark) were compared if stored under lit conditions
– The same or different colours, or levels of light opacity were compared if the oils were stored in the dark.
– The oils were stored at a reasonable (room) temperature for the duration of the study (like it might be stored on a supermarket shelf or a household pantry).
and
3) the time point I chose for discussion was that closest to 12 months, or the latest time point if the study did not extend that long. This was chosen on the basis that 1) EVOO is produced on an annual basis and 2) that if you store bottled EVOO for longer than 12 months you deserve all the rancidity that nature can throw at you (and you probably aren’t reading this blog).
Here is a table that summarises who did what, when, and how.
++++++++++++++++++++++++++++++++++++++++++++++++++++++++++++++++++++++++++++++++++++
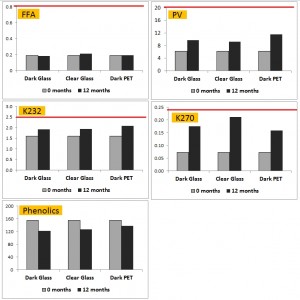
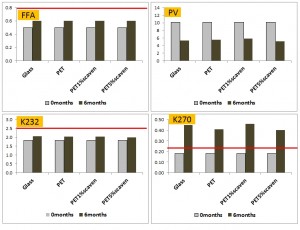
The figure above summarises the recent results of Guillaume and Ravetti (2012). This is the only study that looked at the effect of storing in dark PET under light. The base oil was a good quality, low polyphenol EVOO. Of note was the higher PV in PET (consistent with other studies), but lower measures of secondary oxidation and slightly lower reductions in phenolics compared to dark glass. The dark PET clearly outperformed clear glass on the measures of secondary oxidation. Acidity was largely unchanged by storage in any container type. The red lines show limits for extra virgin status as defined by the International Olive Council (IOC).
++++++++++++++++++++++++++++++++++++++++++++++++++++++++++++++++++++++++++++++++++++++++
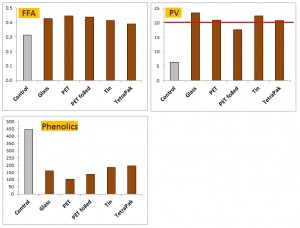
The figure above is compiled from the data of Mendez and Falque (2007). The effect of storage was more strongly felt by this oil than seen in most other studies. The reason for this is unclear. This study also investigated some different packaging types such as tins and tetrapak. The FFA rose in all packaging to around the same extent. Glass produced the highest PV, and PET covered in foil to exclude all light the lowest PV. Losses in polyphenols were greatest in the clear PET, but tetrapak and tins outperformed glass in this respect.
| ++++++++++++++++++++++++++++++++++++++++++++++++++++++++++++++++++++++++++++++++++++++ |
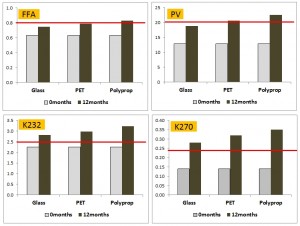
This paper by Pristouri et al. (2009) logically consisted of two parts – the first is glass v plastics and the second compared the different types of PET. I have discussed them separately.
In the first part, polypropylene which has a high oxygen permeability compared to PET was also included. Of note was that a poor quality extra virgin olive oil was used. At the time of packaging it had an FFA of 0.6%, a peroxide close to 10 and most importantly, high levels of secondary and tertiary oxidation. It had all the chemical parameters of a two year old (or more) EU supermarket oil (see the supermarket oil reviews conducted in this blog).
After 12 months, the oil in all packaging types were all above the IOC limits for EVOO for all the parameters measured. In light of the fact that all of them were stuffed, glass outperformed both standard PET and polypropylene, but it was clearly the case of the best of a very bad lot. Despite this, the authors still concluded that PET only had a shelf life of 3 months, and glass 6 months – a conclusion drawn based on the time taken for the already oxidised oil to go beyond the IOC analytical limits. However this experiment is analogous to checking in your 98 year old great-grandfather into a nursing home, and then complaining that the home was to blame when he died the following week!
| +++++++++++++++++++++++++++++++++++++++++++++++++++++++++++++++++++++++++++++++++++++++ |
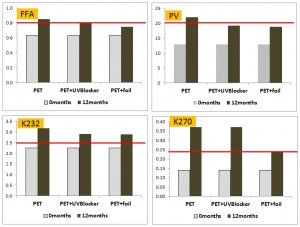
In the second part Pristouri et al. (2009) compared clear PET with foiled covered PET and one containing a UV blocker using the same poor quality oil. Small improvements in performance were seen in the higher tech PET and that covered in foil, but reiterating, this oil was pretty cactus even before it was bottled so I guess it shows that glass is a better container for holding oils on the way to be turned into biodiesel.
| +++++++++++++++++++++++++++++++++++++++++++++++++++++++++++++++++++++++++++++++++++++++ |
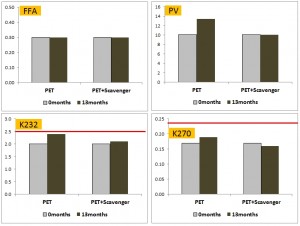
Sacchi et al. (2008) looked at PET v 1.0 and those containing various amounts of oxygen scavengers over a relatively short period of 6 months. The oil stored in all forms of PET oxidised to the same level (chemically) or lower compared to the same oil stored in glass.
This is an important study as it is the first to formally consider the amount of dissolved oxygen in the oil at bottling. The amount of dissolved oxygen at bottling is a crucial variable that is rarely measured. Oxygen is very soluble in olive oil. At saturation, a half litre bottle of olive oil at room temperature will contain 18 milligrams of dissolved oxygen – a level that presumably would not be difficult to achieve if oil is bottled under air (particularly as it has almost half of that level when coming out of the decanter). So what is the relative contribution of PET to oxygen content via transmission during storage? Over 6 months, 3mg of oxygen was transmitted through the walls of a standard 500 mL PET bottle, and 1.5-2.0 mg permeated the walls of PET with oxygen scavengers (di Felice et al. 2008). So based on these figures, the amount of dissolved oxygen that potentially exists in the oil at bottling can far exceed the amount that could potentially be transmitted through through an unopened PET bottle over what is a reasonable amount of time on a shop or pantry shelf. It would also make sense that if an oil has a high level of dissolved oxygen at bottling then the effect of a packaging material with a higher oxygen ingress would have less impact. Other researchers should take note!
Indeed, the authors concluded that “The differences in the shelf life observed between oils bottled in PET and oils bottled in glass are attributable to a difference in the initial content of dissolved oxygen in the oils.”
The tocopherol (a type of Vitamin E which acts as a healthful antioxidant) of the oil was around 190mg/kg at bottling. After 6 months it had dropped to between 25and 40mg/kg with storage in glass experiencing the greatest fall (Figure 2 in their article). These researchers also rated the rancidity of the oils using a sensory panel. The rancidity scores for the oils stored in PET with scavenger were lower (around 1) than when the oil was stored in glass (rancidity score of 3) (Figure 4 in the article).
The researchers also stated the type of capsule that was used to enclose the oil. Glass might be impenetrable to oxygen, but the cap that holds the oil in certainly isn’t, particularly if one uses a polypropylene plastic capsule. No other studies recognised this fact.
| ++++++++++++++++++++++++++++++++++++++++++++++++++++++++++++++++++++++++++++++++++++++++ |
Cecchi et al. (2010) compared standard PET with that containing an oxygen scavenger. A glass treatment was not included in this study, but the results show the improvement in performance of PET containing oxygen scavenging material compared with PET v1.0.
| +++++++++++++++++++++++++++++++++++++++++++++++++++++++++++++++++++++++++++++++++++++++ |
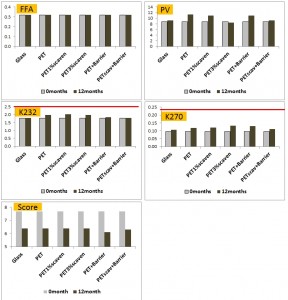
In this study by Gambacorta et al. (2005), the different variants of PET were compared with glass bottles. The trend was for glass being a slightly better container than either PET with scavengers, or with a barrier film. However, when the two technologies were incorporated into the same bottle, the performance of the container was very similar to the glass container both in terms of standard oxidation indices, and sensory quality score. Another aspect worth noting in this study was that the researchers used commercially realistic pack sizes (1L) rather than the often used laboratory sample bottle sizes which do not adequately replicate the volume to surface ratio of commercial packaging.
Conclusion
A review of independently conducted shelf life studies published in referred scientific journals failed to reveal any systematic or significant superiority on the part of glass packaging in terms of standard measures of oxidation when the oils stored in a manner whereby they were exposed to similar amounts of light and at room temperature over 13 months or less.
I’d be very happy to hear from you if you think you see any omissions on the oxidation aspects of using different packaging material, or if you would simply like to discuss my interpretation of the data presented.
Sources:
Cecchi et al. (2010) Study of the quality of extra virgin olive oil stored in PET bottles with or without an oxygen scavenger. Food Chemistry 120, 730–735.
DiFelice et al. (2008) Oxygen permeation in PET bottles with passive and active walls. Packaging Technology and Science 21, 405–415.
Gambacorta et al. (2004) Shelf-life of extra virgin olive oil stored in packages with different barrier properties. Italian Journal of Food Science 16, 417-428.
Guillaume and Ravetti (2012) Evaluation of new analytical methods to detect lower quality olive oils. RIRDC Publication No. 12/007.
Mendez and Falque (2007) Effect of storage time and container type on the quality of extra-virgin olive oil. Food Control 18, 521-529.
Pristouri et al. 2010 Effect of packaging material headspace, oxygen and light transmission, temperature and storage time on quality characteristics of extra virgin olive oil. Food Control 21, 412-418.
Sacchi et al. (2008) Shelf life of vegetable oils bottled in different scavenging polyethyleneterephthalate (PET) containers. Packaging Technology and Science 21, 269–277.
This blog is a personal interpretation of data collected and reported by others. It does not constitute professional advice.







Hi Richard –
Just goes to show how wrong I can be – I thought (or rather, intuited) that plastic would surely protect olive oil less. Any info on other health aspects to be aware of when buying oil in plastic containers (oil being a solvent, BPA, #insert other health buzzwords here#)?
All best,
Tom
[…] Continue reading here: Slick Extra Virgin » Thou shalt not store extra virgin olive oil in plastic … […]
Hi Richard,
Wow! Dude, have you considered a guest appearance on Mythbusters 😉 ?
I want to dig and discuss this more …
Meanwhile, about the concern raised by Tom: there is no BPA in PET plastic: it's mostly in polycarbonate and a few other heavier, stiffer plastics (and, more importantly, lined tin cans). However, PET does leach phthalates and antimony into both drinking water and olive oil (eg here and here). As reviewed here, there is preliminary evidence in vitro and in vivo some combination of these migrants are endocrine disruptors, like BPA — and there is human epidemiological evidence that phthalate exposure increases the risk of obesity and that phthalate exposure increases the risk of reproductive malfunction in males.
Studies do find that phthalates do migrate into olive oil from tin, TetraPak, and even glass containers, but in decreasing order; the phthalates in oils stored in these other containers probably come from liner coatings and (in the case of glass) the plastic caps on the bottles. And, for whatever reason, it appears that olive oil (including EVOO) accumulates higher levels of phthalates than other edible oils.
While not yet conclusive, I think this merits continued avoidance of PET for olive oil storage, irrespective of its effects on oil oxidation. I'm actuallymore concerned about its use for olive oil than the far more common use of PET for soda and drinking water, because of the shorter turnover time and cold storage of the latter.
Hi again Richard,
You’ve emphasized here (and given substantial new weight) a factor that you’ve emphasized before and that is generally neglected: the level of O2 going into the bottle in the first place. Now as you know, there is a rapidly-expanding business model here in the US of olive oil tasting bars, where the oils are stored in stainless steel fusti dispensers; customers go around, sample the oils, and the proprietors bottle up the customer’s selections on the spot.
Now the fustis will have substantial headspace, and the contents can’t really be sparged and it would be very difficult granted regular spouting-out of new oil & its displacement by atomospheric air to keep the headspace topped up with a neutral gas; it’s also going to inevitably lead to substantial dissolved O2 in the freshly-bottled oil.
Aside from trying to minimize or eliminate headspace in the customer’s bottle (either by filling it up to the very edge of the closure, or giving them a squirt of neutral gas), what can these businesses and/or their customers do to to minimize this factor?
Thanks!
Good point Michael
I guess it all gets down to what would be a perfect situation, and how that compares with what is being done right now by the majority.
In the fusti model, people fill up a bottle filled with air with an oil that is probably pretty saturated with oxygen. Oils bottled on bottling lines seem to be very similar, i.e the air in the bottles on the line are displaced with oil that almost certainly contains a lot of dissolved oxygen.
I’m happy to stand corrected on this, but until commercial bottlers start sparging their oils with inert gas to reduce the dissolved oxygen in the oil, and the bottling line is set up to evacuate the bottles before filling and capping them then it seems that those who choose to purchase oils from a fusti are gettng a similar result to the majority of retail oil buyers.
Ideal, no. Practical, probably. Personally as someone who eats a lot of EVOO, I don’t care unless I get lectured by producers about how their oil chemistry is superior to another with respect to theoretical shelf life (emphasis on theoretical), yet they most likely bottle their oxygen saturated oils under air. Condom with a hole in it is an appropriate analogy in these circumstances.
As someone who has been privy to analysis of the volatile composition of various foods and beverages over 20 years, I’m afraid to say that if you are worried about phthalates, then your concern is a bit late. Those f’ers are everywhere! Every gas chromatogram of various foods and beverages that I have ever seen contains distinct peaks that were subsequently assigned to phthalates. Ask the scientist and they just say, “Ah it’s just this phthalate, or that phthalate”.
I’m not saying that they shouldn’t be controlled for the reasons you mention. It’s just that they are, whether we like it or not, universal environmental contaminants. Make anything using plastic hoses, tubes, tanks, bottles, capsule liners (and god knows whatever else), or use wash water that moves through PVC pipes and you will find them.
Yes, I could be accused of being defeatist, but sadly I think that the horse has bolted when it comes to environmental phthalates. Hopefully we will develop some nanotechnology that will turn back the tide one day. But until either that happens, or I unexpectedly join some unknown Amazonian or New Guinean culture, then I won’t feel totally at ease that my average day to day acts of living will not expose me any further phthalates regardless of whether I eat evoo out of PET or not.
Hi Tom. Yes, while writing this post I was completely aware that other factors, both environmental and health, impact on peoples choice of packaging materials, both in general and in EVOO.
I did a brief search of the scientific literature yesterday using the search terms PET and migration. It is a difficult gig as I’m sure that there are other papers around that cover the same topic area which use synonymous keywords. Most of the research has focused on migration of platicisers etc from PET into oil under pretty extreme circumstances, particularly under intensive microwaving.
The one paper I found that measured the migration of oxygen scavengers from PET into oil under typical storage conditions showed that after 60 days, 10mg/kg of the imbedded oxygen scavengers BHT and BHA (in total) migrated into the oil (Tawfik and Huygheart 1999). In my opinion that is 10 mg too much for EVOO, but the addition of up to 200mg/kg of those same antioxidants to refined oils is not unusual. That was just to put things into perspective.
While the research was very solid, I wonder whether the types of PET around in 1999 are the same as today. They may or may not be – commercial in confidence and all makes it difficult to know. There has been a lot of research of late into coating the inside of PET with SiOn (long chain polymers related to glass) in order to prevent migration of this or that.
I wonder myself why not just use glass? I get that, but when you realise that it takes a lot less energy to produce a PET bottle that holds the same volume as a glass bottle (including the energy included in the fossil fuel raw material), and that PET recycling saves 70% compared with the cost of producing the original product while glass pushes 20%, and that PET is substantially lighter resulting in fewer carbon emissions may answer the question.
Some may rightfully point out that recycled PET usually ends up in everything else but new PET bottles (due to contamination with other plastics that limit their reliability to hold pressurised liquids like soda), while glass bottles can be recycled into other glass bottles time and time again. But I’ve been told that there is often an oversupply of green glass which makes it commercially worthless so it gets put into landfill anyway. I only bring all this up to demonstrate that the issues surrounding PET v Glass are pretty complex involving oil chemistry, environmental issues, health and economics. I’m sure whatever argument someone could put up for one over the other, an equally valid counter argument on other grounds could also be put forward. It depends on what is important to you I suppose.
Hi Richard,
Thanks very much. In followup on the phthalate issue: There are, of course, phthalates all over the place; the point is not that there are contaminated and pristine foods, but that the LEVELS vary depending on processing and packaging. The papers I cited, eg, showed variation for specific phthalates and phthalate metabolites, but that altho’ PET varies in migration, PET>TetraPak>Tin>Glass, as you’d expect. And, of course, plenty of foods (such as fresh and even frozen fruits and vegetables) generally have extremely low levels.
To your point about migrants coming out of piping: Casajuana and Lacorte (2003) (one of the studies in the review I cited) “determine[d] target compounds in 7 distribution water and 9 mineral water bottled in polyethylene, polyethyleneter phthalate and glass containers which were analysed upon purchase and after 10 week storage at temperatures up to 30 °C. Distribution water coming from different aquifers which at some point are in contact with plastic or painted concrete reservoirs and pipes, contained dimethylphthalate, diethylphtalate, nonylphenol, buthylbenzylphthtalate and DEHP at concentrations ranging from 0.005 to 0.331 μgL−1, depending on the sampled crea whereas bottled water showed levels up to 1.7 μg L−1 of some of the studied compounds, attributed to 10 week storage conditions. ” The review says in particular that this study found that “phthalates in water from glass bottles were still generally undetectable after 10 weeks of storage. However, in their samples of water from PET bottles, three out of five brands showed measurable levels of di-(2-ethylhexyl) phthalate (DEHP) after 10 weeks of incubation, with an average DEHP concentration of 0.134 µg/L, and all five brands showed measurable levels of DEP after 10 weeks of incubation, with an average DEP concentration of 0.214 µg/L.”
The human epidemiology (if it is not actually reflecting a non-causal association) and the experimental work certainly indicate that there is a dose-response effect on adverse outcomes, so the idea is to minimize exposure. Here eg. is a guide on how to greatly reduce exposure to bisphenol A. There has been a lot less focus on phthalates to date, but I’m sure that a similar guide could be written (and, indeed, would contain many or most of the same suggestions: less conventional animal products, less animal fat, less consumption of liquids stored for extended periods in the relevant plastic types, more fresh and frozen produce.
And, apparently, olive oil from glass containers.
This also should lead, generally, to less of these substances getting out into the environment, where they certainly seem to be wreaking havoc on various amphibians, and possibly other creatures.
I do, certainly, take your point about the carbon advantages of plastic. Fortunately, companies are working to develop truly non-endocrine-disrupting plastics in response to the bad press on BPA.
Costco sells 2 liter bottles of extra virgin olive oil packaged in dark green PET. I buy it because it’s half the cost of EVOO anywhere else, and it’s good. I wonder if I should be decanting it into smaller opaque glass bottles and storing them in the freezer. On the other hand, I go through about two liters/month, so the storage time in my house versus time in the Costco supply line is pretty minimal. Right now I decant it one liter at a time into a smaller glass bottle that sits on the counter, and the other half stays in the dark pantry cupboard until I need it. I do worry about the pthalates. I keep canned food to a minimum and use glass for all my leftover and food storage, so buying olive oil in plastic is a significant departure from policy for me.